כנס החברה הגיאולוגית 2014
כנס החברה הגיאולוגית 2014
השנה, במסגרת כנס החברה הגיאולוגית הישראלית שנערך במצפה רמון, ניתנו שתי הרצאות של יועצי החברה הבינ"ל מאוניברסיטת מק'וורי שבסידני, אוסטרליה וזאת כחלק ממושב "גיאולוגיה בתעשייה ומחצבים-חומרי גלם בישראל".
הרצאתו של פרופ' ביל גריפין (Professor William L. Griffin) השנה התמקדה בהמשך המחקר המינרולוגי-גיאוכימי המתנהל על ידו באמצעות מינרלי הקורונדום אותם מאתרת שפע ימים הן במקורות הראשוניים והן במקורות המישניים בשטחי ההיתרים בהם החברה פועלת. פרופ' גריפין ושותפיו למחקר מתארים מערכת געשית (וולקנית) מסועפת, כעין "צנרת" בעומק עשרות קילומטרים מתחת להרי הגעש בכרמל ובגליל. מאסף המינרלים הללו הוא תשתית להבנת עומק היצירה של המינרלים. יצויין כי המינרלים הייחודיים בתוך גבישי הקורונדום מתגלים בעזרת מיקרוסקופיה משוכללת בתנאי מעבדה.
חוקרת נוספת מהצוות האוסטרלי בשם Dr. Jin-Xiang HUANG תיארה את המויסאנייט (SiC) ואת הפרטים הייחודיים המקשרים בין המויסאניטים שנתגלו על ידי שפע ימים בישראל לבין אזורים אחרים בעולם. המויסאנייט מישראל לדבריה, מאפשר חלון מרתק למעמקי המעטפת, אל אזורים בהם החמצן דליל באופן קיצוני והטמפרטורות גבוהות ביותר (עד כדי 1375 מעלות צלסיוס!!). המערכות הגעשיות הללו עשירות במתאן ובפחמן. אלו סביבות שמוכרות לנו רק ממערכת השמש (כלומר מהחלל החיצון) ואילו כאן, באמצעות המינרלים שהתגלו על ידי שפע ימים ניתן לחדש למדע אודות היווצרות כדור הארץ הקדום, על בסיס אלפי גבישי המויסאנייט הטבעיים המאותרים בשטחי ההיתרים בהם פועלת החברה.
גביש המויאסנייט הגדול ביותר שהתגלה עד כה בעולם הינו בגודל של 4.14 מ"מ והוא נמצא ע"י שפע ימים (12/2015) במדגם שנאסף במקטע התיכון של נחל הקישון. יצויין כי ממצא זה מצטרף ל- 2 ממצאים נוספים קודמים בגדלי WORLD RECORD"" של 4.1 מ"מ (08/2012) ו- 3.5 מ"מ (03/2009).
לאחר ההרצאות, החברה הציגה מידע משלים במושב הפוסטרים, יחד עם יועץ האקספלורציה ד"ר ג'ון ווארד ועם נציגים נוספים מהצוות האוסטרלי: פרופ' סו או'ריילי (Professor Sue O'Reilley) והחוקרת שרה גיין (Sarah Gain).
פרופ' גריפין (Prof. William L. Griffin,) מסכם את ביקור העבודה האחרון שלו (מרץ, 2017) בשטחי האקספלורציה ובמתחם התפעולי של החברה בעכו
פרופ' גריפין הינו חוקר מוביל בתחום חקר מעטפת כדור הארץ העמוק, המנהל את המרכז לחקר האבולוציה הגיאוכימית והמטלוגנזה (תהליכי יצירת מתכות) של היבשות (GEMOC) באוניברסיטת מק'קוורי (Macquarie) שבסידני, אוסטרליה. עוסק בגיאוכימיה, גיאופיסיקה ובפטרולוגיה של הקרום התחתון ושל אזור המעבר בין הקרום למעטפת כדור הארץ. מפתח יישומים ייחודיים לחקר מיקרוסקופי של יסוד קורט (יסודות נדירים על פני כדור הארץ שגודלם וכמותם מזעריים והם בעלי השלכות לגבי הסביבה הכימית הדינאמית בה אנו חיים). מומחה בחיפוש והתוויית אזורי מטרה עבור תעשיית המינרלים. בנוסף, היה עורך של מגזינים וספרות מקצועית, קיבל מדליות ופרסי כבוד עבור מחקריו הן בארצו והן בכמה אוניברסיטאות בעולם.
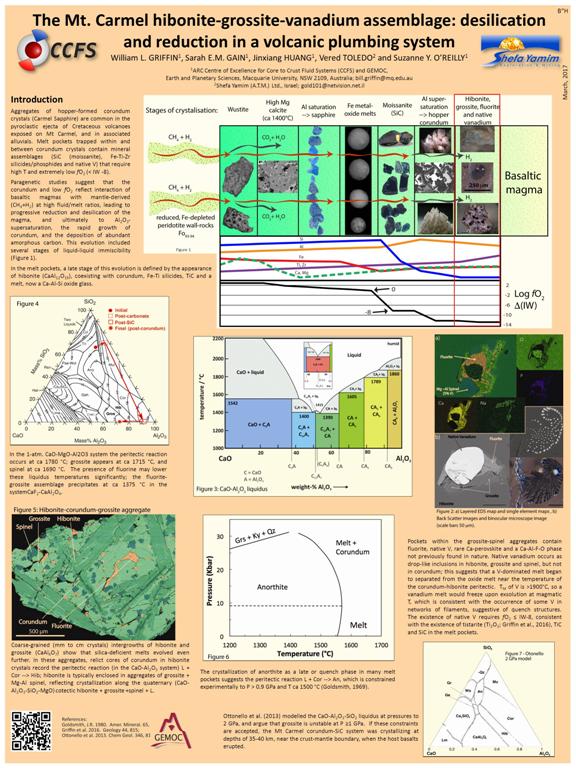
The hibonite-grossite-vanadium assemblage in Mt Carmel corundum: extreme desilication and reduction in a volcanic plumbing system
William L. GRIFFIN, Sarah E.M. GAIN, Jinxiang HUANG, Vered TOLEDO and Suzanne Y. O’REILLY
Aggregates of corundum crystals (Carmel Sapphire) are common in the pyroclastic ejecta of Cretaceous volcanoes exposed on Mt Carmel (Lower Galilee). Melt pockets trapped within and between corundum crystals contain mineral assemblages) that require high T and extremely low fO2 (IW -10). Paragenetic studies suggest that the corundum and low- fO2 reflect interaction of basaltic magmas with mantle-derived (CH4+H2) at high fluid/melt ratios, leading to progressive reduction and desilication of the magma, inducing Al2O3-supersaturation, the rapid growth of corundum, and the deposition of amorphous carbon.
In the melt pockets, a late stage of this evolution is defined by the appearance of hibonite (CaAl12O19), coexisting with corundum, Fe-Ti silicides, TiC and a Ca-Al-Si oxide glass. However, the occurrence of coarse-grained (mm to cm crystals) intergrowths of hibonite and grossite (CaAl4O7) shows that silica-deficient melts evolved even further. In these aggregates, relict cores of corundum in hibonite crystals record the peritectic reaction L + Cor Hib; hibonite is typically enclosed in aggregates of grossite + Mg-Al spinel, reflecting crystallization along the quaternary (CaO-Al2O3-SiO2-MgO) cotectic hibonite + grossite +spinel + L. Pockets within the aggregates contain fluorite, native V, Ca-perovskite and a Ca-Al-F-O phase not previously found in nature. Native vanadium occurs as drop-like inclusions in hibonite, grossite and spinel, but not in corundum; this suggests that a melt dominated by V began to separate from the oxide melt near the temperature of the corundum-hibonite peritectic reaction. The melting point of V is >1900 °C, so that a vanadium melt would freeze upon exsolution at magmatic T, which is consistent with the occurrence of some V in networks of filaments, suggestive of quench structures. The existence of native V requires fO2 ≤ IW-8, consistent with the existence of tistarite (Ti2O3; Griffin et al., 2016 Geology 44, 815), TiC and SiC in the melt pockets.
In the 1-atm. CaO-MgO-Al2O3 system the peritectic reaction occurs at ca 1780 °C; grossite appears at ca 1715 °C, and spinel at ca 1690 °C. The presence of fluorine may lower these liquidus temperatures significantly; fluorite+grossite precipitates at ca 1375 °C in the system CaF2-CaAl2O4. The crystallization of anorthite as a late or quench phase in many melt pockets suggests the peritectic reaction L + Cor An, constrained experimentally to P > 0.9 GPa and T ca 1500 °C (Goldsmith, 1980, Amer. Mineral. 65, 272). Modelling by Ottonello et al. (2013 Chem Geol. 346, 81.) suggests that grossite is no longer on the liquidus at P ≥1 GPa. If these constraints are accepted, the Mt Carmel corundum system was crystallizing at depths of 35-40 km, near the crust-mantle boundary, when the host basalts erupted.
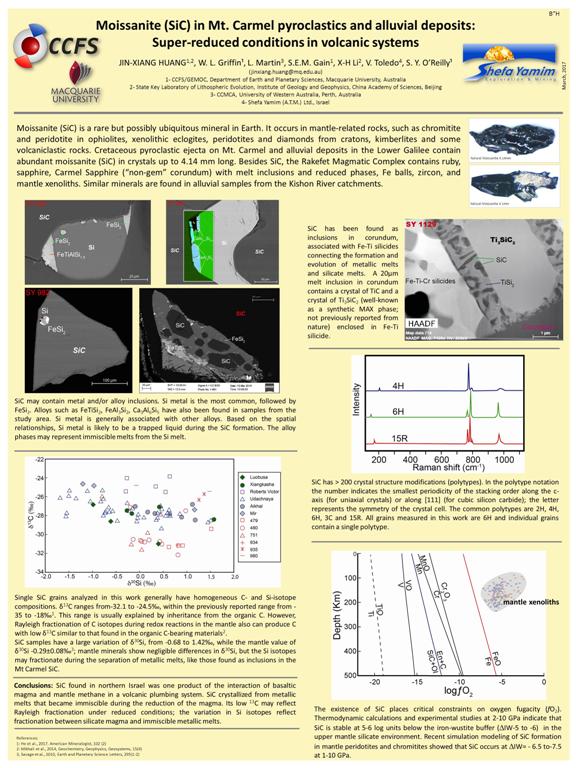
Moissanite (SiC) in Mt Carmel pyroclastics and Kishon River alluvial deposits: Super-reduced conditions in volcanic systems
Jin-Xiang HUANG, William L. GRIFFIN, Sarah E.M. GAIN, Vered TOLEDO and Suzanne Y. O’REILLY
A striking feature of Cretaceous pyroclastic ejecta on Mt Carmel and Kishon river alluvial deposits in the Lower Galilee is the presence of abundant moissanite (SiC) in crystals up to 4.41 mm long. Experimental data and theoretical calculations suggest that SiC is only stable at log ƒO2 ≤(∆IW-6.5) at upper mantle conditions. Its occurrence in the volcanic products indicates that either the volcanic system experienced a reduced stage or that at least parts of the wall rocks of the volcanic conduit contained reduced minerals.
Moissanite is a highly covalent compound of Si and C in a ratio of 1:1 in hexagonal-cubic crystal system. It is rarely found in terrestrial rocks, but has been reported in diamonds, kimberlites, ophiolites, eclogites, serpentinites and xenolithic carbonatites; the natural occurrence of some of these has been debated. However, it is the best-studied type of presolar grain, providing critical information on nucleosynthesis and stellar evolution.
Moissanite can have more than 200 polytypes. SiC grains from Mt Carmel and other terrestrial rocks are mainly the 6H polytype; a few are the 4H and 15R polytypes. The polytype of the grain does not have any obvious correlation with its petrographic or geochemical characteristics.
Moissanite from Mt Carmel may contain inclusions, such as native Si, Fe-, CaAl-, FeAl-, FeTi-silicide and TiV, similar to those reported from kimberlitic and ophiolitic SiC. The occurrence of such alloy inclusions indicates that they were formed in reduced conditions: log ƒO2 <∆IW-8.
Mt Carmel moissanite and Ti-, Fe-silicides are also found as inclusions in corundum (Carmel Sapphire) with melt pockets containing reduced phases. Paragenetic studies suggest that the corundum and low fO2 reflect interaction of mafic magmas with mantle-derived (CH4+H2) at high fluid/melt ratios, near the crust-mantle boundary leading to progressive reduction and desilication of the magma, and ultimately to Al2O3-supersaturation, the rapid growth of corundum, and the deposition of abundant amorphous carbon.
The Si- and C-isotope compositions of Mt Carmel moissanite were analyzed in-situ by SIMS (CAMECA 1280). The data show a large variation in both δ30Si (-0.68 to +1.42 ‰) and δ13C (-25 to -32‰) among different grains. Individual grains generally have homogeneous isotopic compositions. Such variation is not related to differences in polytypes or the occurrence of inclusions, suggesting it may be related to heterogeneity of the source(s) or unidentified fractionation processes that operated during crystallization.
Moissanite from this area is highly likely to be a direct volcanic product of the interaction of CH4+H2 with the basaltic magma. However, it cannot be completely excluded that some of the grains are xenocrysts from the mantle, which were formed by other processes such as mantle metasomatism by reduced fluids.
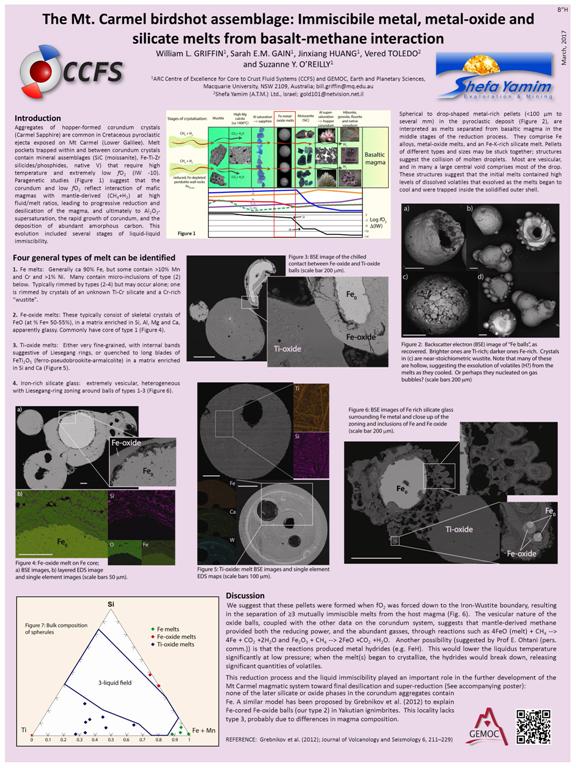
The Mt. Carmel birdshot assemblage: Immiscibility of metal, metal-oxide and silicate melts during basalt-methane interaction in a volcanic plumbing system
William L. GRIFFIN, Sarah E.M. GAIN, Jinxiang HUANG, Vered TOLEDO and Suzanne Y. O’REILLY
Aggregates of corundum crystals (Carmel Sapphire) occur in Cretaceous pyroclastic ejecta on Mt Carmel (Lower Galilee). Melt pockets trapped within and between corundum crystals contain mineral assemblages that require high T and extremely low fO2 (IW -10). Paragenetic studies suggest that the corundum and low fO2 reflect interaction of mafic magmas with mantle-derived (CH4+H2), leading to reduction and desilication of the magma, inducing Al2O3-supersaturation, the rapid growth of corundum, and deposition of amorphous carbon.
Spherical to drop-shaped metal-rich pellets <100 µm to several mm in diameter are common in the pyroclastics, and are interpreted as melts separated from basaltic magma. Four general types of melt can be identified.
1. Fe melts: Generally ca 90% Fe, but some contain much higher Mn, Cr and Ni). Many contain micro-inclusions of type (2) below. Typically rimmed by types (2-4).
2. Fe-oxide melts: Example: SiO2 6%, TiO2 2%, Al2O3 2%, MgO 1%, FeO 87% CaO 1.5 %. These typically consist of skeletal crystals of stoichiometric FeO (at % Fe= 50-55%), in a matrix enriched in Si, Al, Mg and Ca. Commonly have core of type 1.
3. Ti-oxide melts: Either very fine-grained quenched to long blades of FeTi2O5 in a matrix enriched in Si and Ca. Examples: (1) SiO2 10%, TiO2 55%, Al2O3 3%, MgO 2%, FeO 20 %, MnO 7%, CaO 3%; (2) SiO2 1%, TiO2 69%, Al2O3 0.3%, FeO 14%, MnO 12%, CaO 0.5%, WO 2%.
4. Iron-rich silicate glass: extremely vesicular, heterogeneous with Liesegang-ring zoning around balls of types 1-3. Mean composition SiO2 40%, TiO2 1%, FeO 30%, MnO 11%, Na2O 2%, K2O 14%.
Pellets of different types and sizes may be stuck together. Most droplets are vesicular, and in many a large central void comprises most of the drop. These structures suggest that the melts contained high levels of volatiles that exsolved as the melts began to cool.
We suggest that these pellets formed when fO2 dropped to the Iron-Wustite boundary, resulting in the separation of mutually immiscible melts from the host magma. The vesicular nature of the oxide balls, coupled with other data on the corundum system, suggests that mantle-derived methane provided both the reducing power, and the abundant gasses, through reactions such as 4FeO (melt) + CH4 4Fe + CO2 +2H2O and Fe2O3 + CH4 2FeO +CO2 +H2O. This immiscibility played an important role in the further development of the Mt Carmel magmatic system: none of the silicate or oxide phases in the corundum aggregates contain Fe, because most of it had been removed earlier. A similar model has been proposed by Grebnikov et al. (2012; Journal of Volcanology and Seismology 6) to explain Fe-cored Fe-oxide balls (type 2) in Yakutian ignimbrites.
צרו קשר
Thank you for contacting us.
We will get back to you as soon as possible
We will get back to you as soon as possible
Oops, there was an error sending your message.
Please try again later
Please try again later
שפע ימים בע"מ
כתובת:
רחוב הרצל 90, ת.ד. 720, נתניה - 4210602
טלפון:
09-8331999
פקס:
09-8619171
דוא"ל:
info@shefayamim.com
נבנה ע"י קידום פלוס בניית אתרים
